 When a system, for example, n moles of a gas of volume V at pressure p and temperature T, is created or brought to its present state from absolute zero, energy must be supplied equal to its internal energy U plus PV, where PV is the work done in pushing against the ambient (atmospheric) pressure. The U term can be interpreted as the energy required to create the system and the PV term as the work that would be required to "make room" for the system if the pressure of the environment remained constant. In practice, a change in enthalpy (dH) is the preferred expression for measurements at constant pressure, because it simplifies the description of energy transfer. 
The total enthalpy of a system cannot be measured directly, because the internal energy contains components that are unknown. H is enthalpy, U is internal energy, P is pressure and V is volume. Hydrogen gas will have higher entropy than liquid water.Įnthalpy is defined as the sum of the system's internal energy and the product of its pressure and volume, dH = dU + PdV. This is because gas molecules are widely spread out and, therefore, are more disordered than solids and liquids. Entropy, or the amount of disorder, is always highest for gases and lowest for solids. 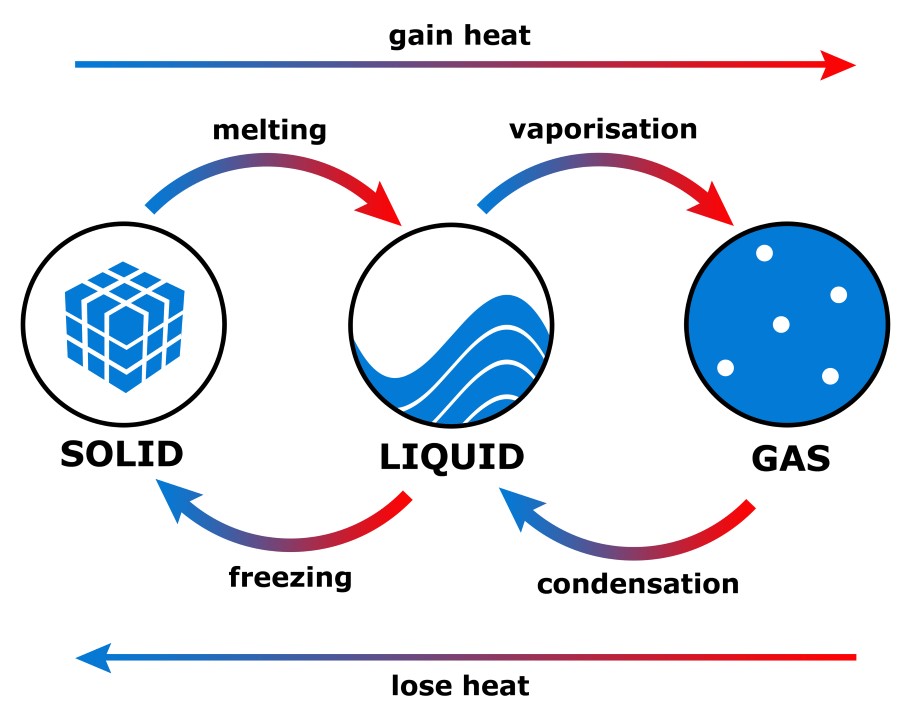
Enthalpy is zero for elemental compounds such as hydrogen gas and oxygen gas therefore, enthalpy is nonzero for water (regardless of phase). Enthalpy is the amount of internal energy contained in a compound whereas entropy is the amount of intrinsic disorder within the compound. 
0 Comments
Leave a Reply. |
Details
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
